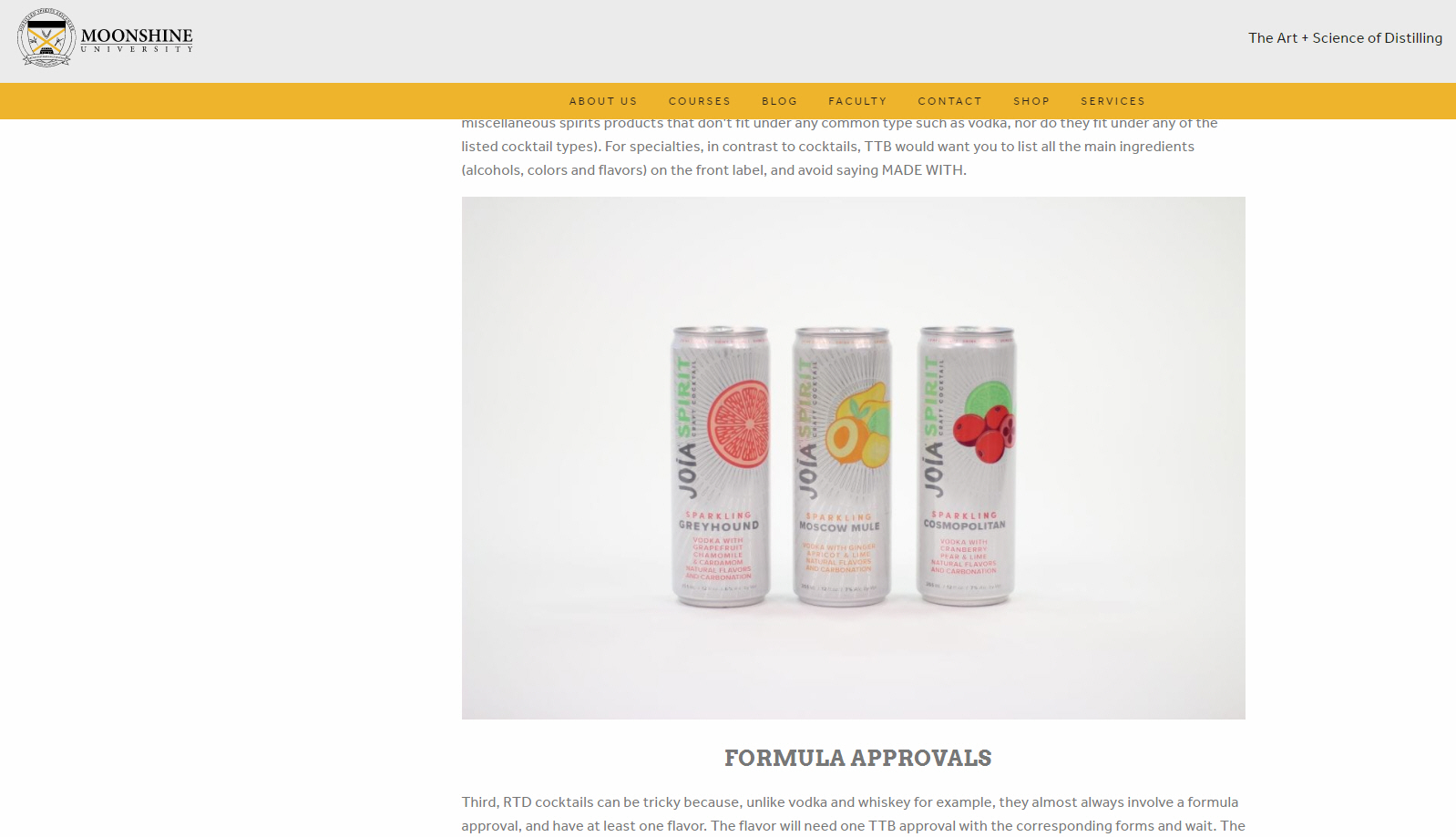
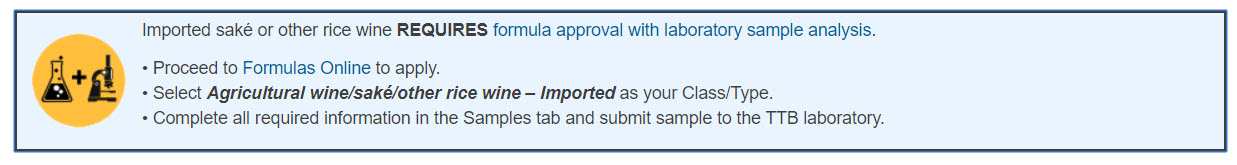
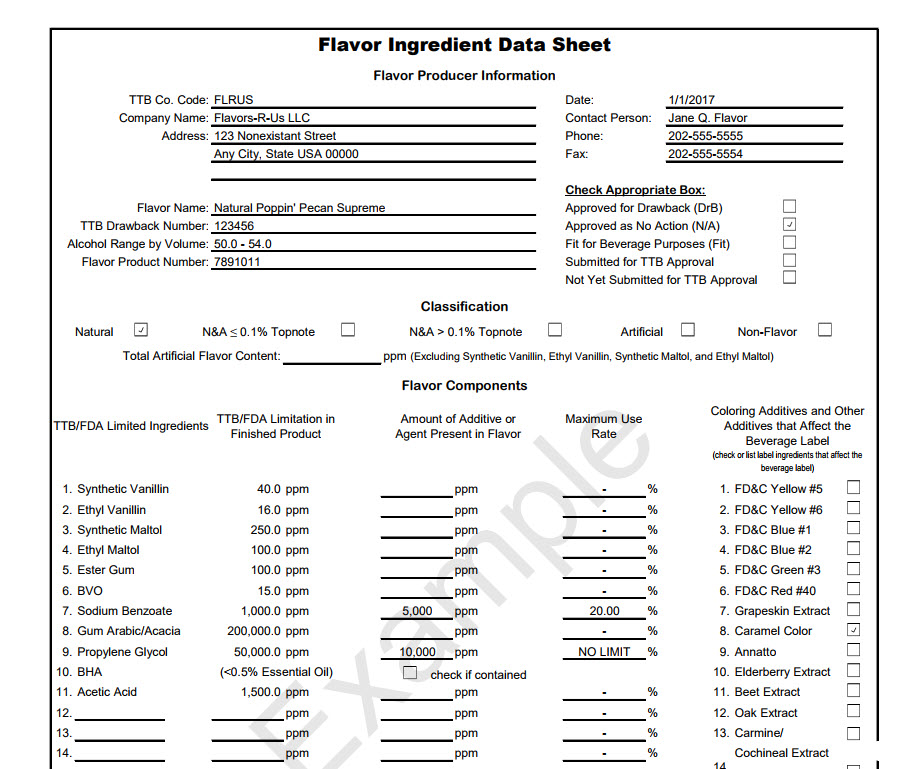
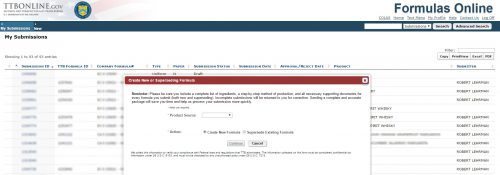
Join Marshall’s talk on TTB label and formula issues, coming up soon. This event is hosted by the Brewers Association on January 13, for BA Members only. The sign-up details are here and the webinar will cover the following:
In this BA Collab Hour webinar, we will discuss common mistakes made during the Alcohol and Tobacco Tax and Trade Bureau (TTB) label and formula submission processes, and go over some tips and (compliant) tricks that may make your submissions and interactions with TTB a less frustrating process.
The webinar will focus largely on beer, seltzer and malt beverage issues. Marshall is drawing on experience from thousands of label and formula approvals (as well as “a few” rejections and needs corrections) handled by himself and his colleagues, and most attendees should learn at least a few practical tips along the way.
Continue Reading Leave a Comment